Periodic Table of Elements - Rubidium - Rb. 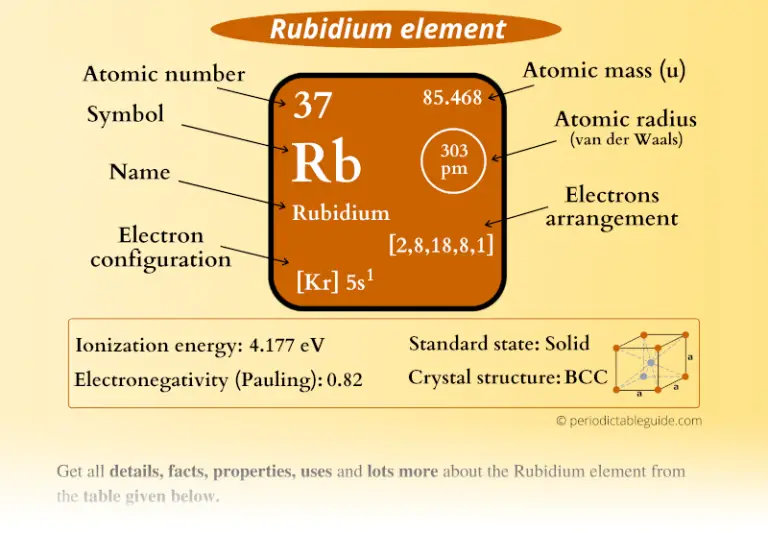
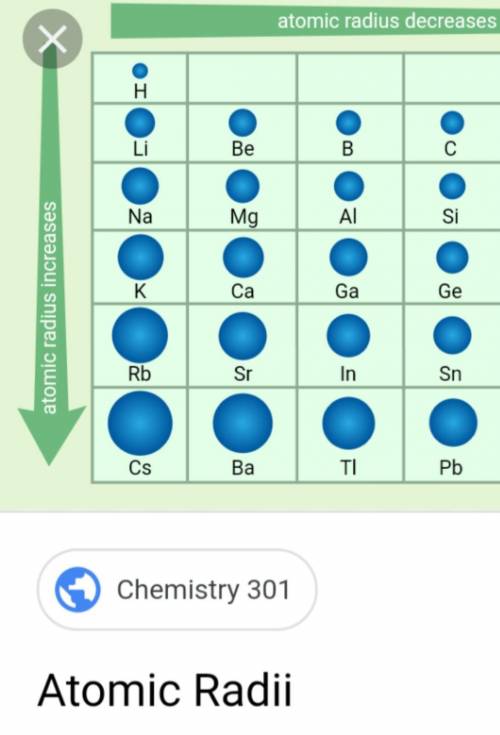
If you need to cite this page, you can copy this text: This database focuses on the most common chemical compounds used in the home and industry. Molar mass calculations are explained and there is a JavaScript calculator to aid calculations. Molar Mass Calculations and Javascript Calculator.Introduces stoichiometry and explains the differences between molarity, molality and normality. Related ResourcesĪnswers many questions regarding the structure of atoms. Uses of Rubidium: Has limited commercial uses and is primarily used for research purposes.Ī list of reference sources used to compile the data provided on our periodic table of elements can be found on the main periodic table page.Usually obtained from lithium production. Sources of Rubidium: Occurs abundantly, but so widespread that production is limited.Name Origin: Latin: rubidus (red) the color its salts impart to flames.Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances. Vapor Pressure = C Regulatory / Health.Enthalpy of Atomization: 82 kJ/mole 25☌.Description: Soft silver white metal that oxidises rapidly and ignites in air.Conductivity Electrical: 0.0779 10 6/cm Ω.Coefficient of lineal thermal expansion/K -1: 90E -6.Valence Electron Potential (-eV): 9.47 Physical Properties of Rubidium.Electronegativity: 0.82 (Pauling) 0.89 (Allrod Rochow).Electrochemical Equivalent: 3.1888g/amp-hr.Valence Electrons: 5s 1 Electron Dot Model.Number of Neutrons (most common/stable nuclide): 48.Number of Electrons (with no charge): 37.Electrons per Energy Level: 2,8,18,8,1 Shell Model.Electron Configuration: 1s 2 2s 2p 6 3s 2p 6d 10 4s 2p 6 5s 1.Crystal Structure: Cubic body centered.Atomic Mass 85,4678 Learn more about the atomic mass. Rubidium Data Rubidium Atomic Radius 2.98 Å State at 20 ☌ Solid Uses Used as a catalyst, photocells, and vacuum and cathode-ray tubes. There are cool facts about Rubidium that most dont know about. Cross Section (Thermal Neutron Capture) σ a/ barns : 0.38 In the case of Rubidium the atomic radius is 2.98 Å.Swedish: Rubidium Atomic Structure of Rubidium.Series: Alkali Metals Rubidium's Name in Other Languages.Potential Parent Nuclides Overview of Rubidium.In addition technical terms are linked to their definitions and the menu contains links to related articles that are a great aid in one's studies. Common chemical compounds are also provided for many elements. Which has the smallest radius K Rb K Rb Potassiums atomic radius is smaller than. Skip to site menu on this page Periodic Table of Elements Element Rubidium - RbĬomprehensive data on the chemical element Rubidium is provided on this page including scores of properties, element names in many languages, most known nuclides of Rubidium. Since it is easily vaporized, rubidiums spectral absorption range is a good target for laser manipulation of atoms. What is the atomic radius of Rb Rubidium has an atomic radius of 248 picometers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
